Novel Cellulose-Based Drug Nanocarrier Can Help Overcome Bacterial Resistance in Eye Infections
Bacterial eye infections, technically known as bacterial keratitis, remain one of the main reasons for vision loss throughout the world. Despite the wide availability of antibiotic eye drops to treat bacterial keratitis, microbes often develop mechanisms that grant them resistance—making conventional treatments ineffective. For example, some species of bacteria can surround themselves in a network of proteins and DNA, called ‘biofilm,’ that limits how deep molecular drugs can penetrate their populations. Now, a team of multidisciplinary researchers from the Birla Institute of Technology and Science (BITS)-Pilani, Hyderabad Campus and the Indian Institute of Technology (IIT) Delhi, both in India, have crafted a new drug delivery system that can pierce through this bacterial armour, empowering a conventional antibiotic typically used for bacterial keratitis. Their findings are published in Carbohydrate Polymers.
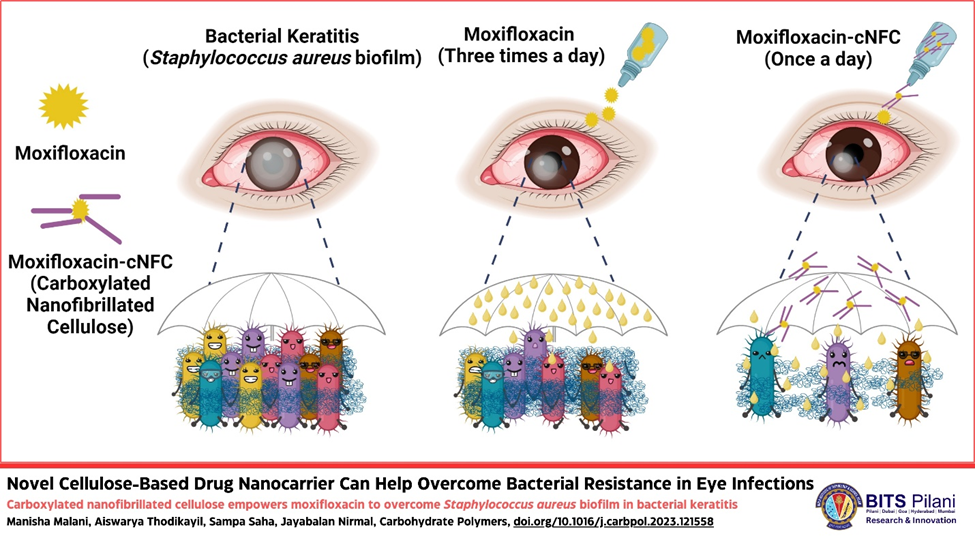
The antibiotic in question is moxifloxacin (Mox). Although previous studies have shown that this compound can kill Staphylococcus aureus, a bacterium responsible for many corneal infections, it is unable to penetrate indwelling bacteria surrounded by biofilm. To overcome this issue, the research team—which was led by Prof. Nirmal Jayabalan and Prof. Sampa Saha, along with their Ph.D. Scholars Manisha and Aiswarya, from the Translational Pharmaceutics Research Laboratory at the Department of Pharmacy, BITS-Pilani, Hyderabad Campus, and the Particle and Surface Engineering Laboratory at the Department of Material Science and Engineering, IIT Delhi—resorted to a natural nanomaterial: cellulose nanofibrils. Their goal was to modify these nanofibrils appropriately to use them as a vehicle for Mox, making sure it reaches its intended targets. “The presence of multiple hydroxy groups (-OH) on cellulose allows for its chemical modification, thus lending itself for the development of materials with adaptive features,” explains Dr. Nirmal and Dr. Sampa.
The research employed a two-step chemical modification process that replaced the hydroxy groups with carboxyl groups (-COOH), producing carboxylated nanocellulose fibres (or ‘cNFCs’). The exposed carboxyl groups can adsorb Mox through both electrostatic interactions and hydrogen bonds, producing Mox-cNFCs. It is worth noting that the carboxyl groups also contribute to the dispersibility and penetration of cNFCs, as demonstrated in previous studies.
Characterisation analyses showed that the proposed strategy could successfully produce Mox-cNFCs with a high drug load. More importantly, these drug carriers were able to release their payload in a controlled fashion over a long period of time, which is an improvement over standard Mox eye drops. “Mox-cNFCs provide an opportunity to sustain anti-bacterial effects for prolonged periods, which makes them more efficient in combating infections,” remarks Dr. Nirmal. Simply put, by using the proposed drug carrier, patients would need to apply antibiotic eye drops once per day rather than multiple times a day. “From the patient's perspective, taking multiple eye drops until the infection recedes without missing a single dose is challenging, and failing to do so can result in patients losing their gift of vision in severe cases,” adds Dr. Nirmal. “Our therapeutic approach could improve patients' quality of life significantly due to the reduced frequency of eye drop administration, and also accelerate vision recovery by clearing bacterial loads faster.”
Through in vitro experiments using bacterial cultures, the researchers showed that Mox-cNFCs could disrupt the biofilm matrix, realising higher drug penetration and efficacy without an increase being needed in the Mox dose. Afterwards, the team conducted in vivo experiments in rats with ocular infections to assess the effectiveness of the proposed drug delivery system. Whereas plain Mox only cleared infections when applied three times daily, Mox-cNFC could resolve bacterial keratitis with a once-a-day application protocol—thanks to its slow, sustained drug release and its high adhesion to pre-corneal tissue.
Overall, this innovative strategy will not only serve in the fight against bacterial keratitis, but also help tackle the ever-growing threat of antibiotic-resistant bacteria. By empowering antibiotics such as Mox using drug delivery nanotechnology to overcome bacteria’s defence mechanisms, the amount of antibiotics required per patient will decrease. These efforts are in line with other research projects currently under development at BITS-Pilani, as Dr. Nirmal comments: “Our team at the Hyderabad campus is developing several platform-based technologies such as nanomicelles, ocular polymeric patches, microspheres, stimuli-responsive gel and other delivery systems, which have the potential to be translated from the lab bench to the clinic.” His team's central focus is to achieve “Optimum Therapeutics” to the eye by using these efficient platform carrier systems to deliver drugs at the right place and time, and in the right amount. He also notes that continuous efforts between academic researchers, clinicians, and pharmaceutical companies are essential for translating new treatment modalities from the lab bench to the clinic. With this vision in mind, the Translational Pharmaceutics Research Laboratory (TPRL) has been established.
Indeed, the fight against antimicrobial resistance is a collective responsibility, and not just for individuals alone. Each person can contribute to preventing the rise of resistance in multiple ways. At BITS-Pilani, researchers are actively engaged not only in advancing research but also in addressing societal concerns. “We annually organise the 'Responsible Medicine Disposal Program' to educate society about the rise of antimicrobial resistance due to careless disposal of medicine,” says Dr. Nirmal. “Along with raising awareness, we collect unused and expired medicines from door to door and ensure their proper disposal to reduce the burden of antibiotics on planet Earth.”
Let us hope that translational science, heightened awareness, and widespread cooperation pave the way to a healthier future.