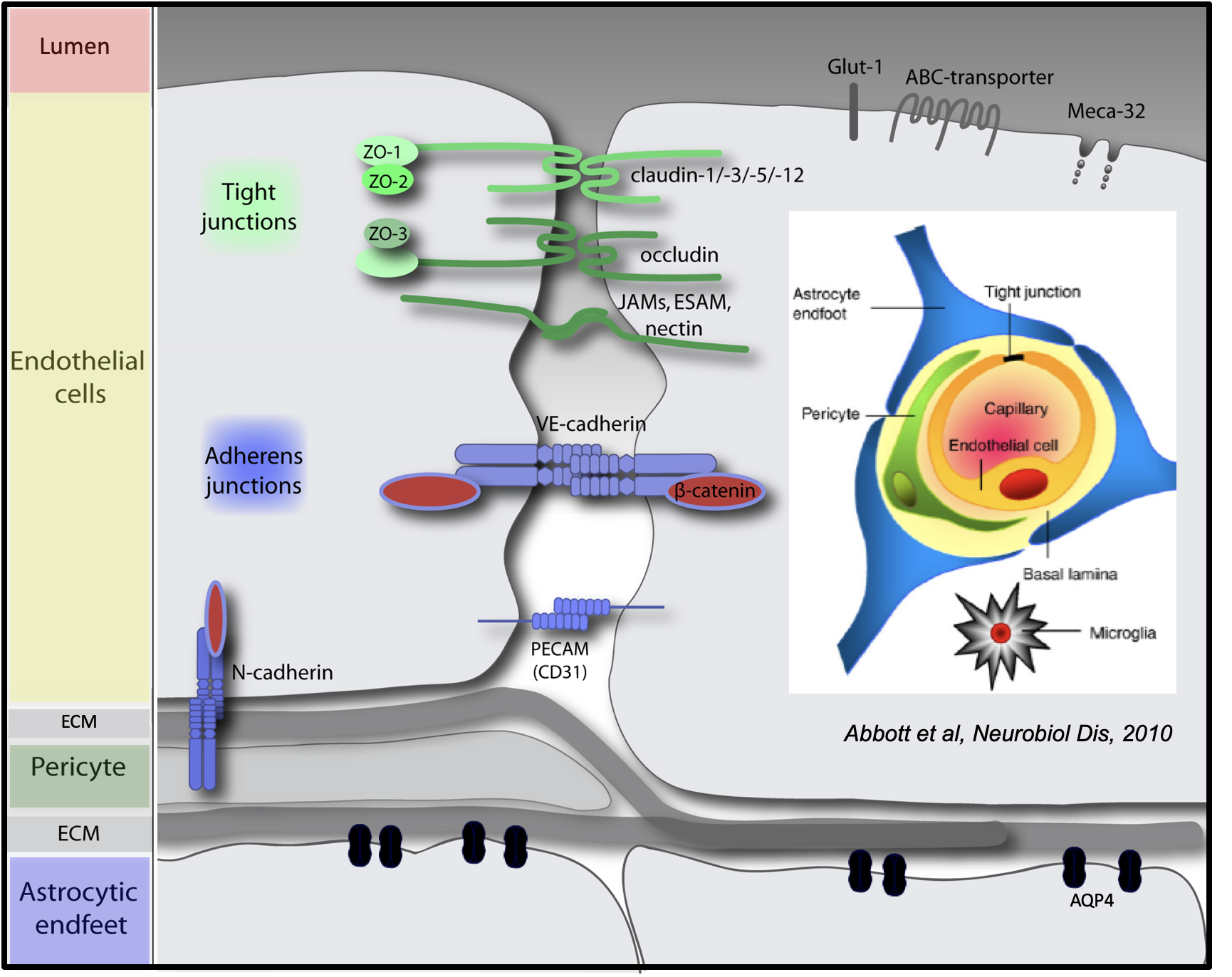
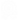Professional Career
2001-2002 Teaching Assistant, Biomedical Engg., Worcester Polytechnic Institute WPI, USA
2001-2001 Summer Intern, Genzyme Inc. (Sanofi), Framingham, MA, USA
2002-2002 ADA Summer Intern, Neural & Behavioral Sciences, Penn State Univ, PA, USA
2003-2003 COSAT Summer Fellow, Neutrogena (Johnson & Johnson company), LA, USA
2004-2009 Research Assistant, Neural & Behavioral Sciences, Penn State University, PA, USA
2006-2007 Graduate Lecturer: Penn State University, Hershey, PA, USA
2010-2015 Postdoctoral Fellow, Institute of Neurology, Goethe University, Frankfurt, Germany
2015-2018 Group Leader, Pharmazentrum & Edinger Institute, Goethe University, Frankfurt, Germany
2019-2023 Group Leader (Associate Professor), Edinger Institute, Goethe University, Germany
2023-current Associate Professor, BITS-Pilani, Hyderabad, India